Google Ads
Module 1.0
Atomic Structure of Matter
- After studying this section, you should be able to:
- Describe the basic constituents of Matter.
- •Elements, Mixtures, Compounds, Atoms, Ions and Molecules.
- Describe a Silicon Atom.
- • Schematic representation.
- • Distribution of protons and electrons.
- Describe the electrical properties of materials.
- • Conductors, insulators and semiconductors.
- Describe the effects of temperature on conductors and semiconductors.
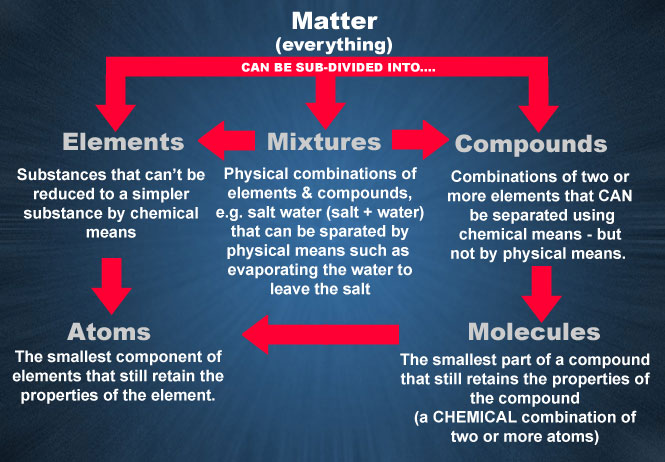
Fig.1.0.1 Sub-divisions of Matter.
Matter is anything that occupies space and has weight. It may exist in any one of three states: solid, liquid or gas. Matter includes the air we breathe, the food we eat, all the objects around us and even ourselves. Since matter includes so much, it is better to divide matter into some sub divisions as shown in Fig. 1.0.1.
Elements Mixtures and Compounds
An element is a basic building block of matter. It is a substance that can't be reduced to a simpler state by chemical means. There are 103 known elements, including 16 man made elements. Examples are: copper, gold, silver, hydrogen and silicon.
When two or more elements are combined, the combination is called a compound. Compounds are formed when two or more atoms share part of their structure. They can be separated by chemical means, such as distillation but not by any physical means. Examples of compounds are: water (hydrogen + oxygen) and salt (sodium + chlorine). The smallest part of a compound which still retains the properties of the compounds (the combination of atoms) is called a molecule. A molecule is the chemical combination of two or more atoms, and an atom is the smallest part of a single element which retains the properties of that element.
When elements and compounds are combined they are called a mixture. Examples of mixtures include air, (oxygen + nitrogen + carbon dioxide and other gases) and salt water (salt + water).
Atoms
Atoms are characteristic of particular elements, so as there are over 100 known elements, then there are over 100 different known atoms
Every atom has a nucleus located at the centre of the atom. In nearly all atoms the nucleus contains two types of particles called protons and neutrons. The neutrons have a neutral electric charge (no charge). Protons each have a positive electric charge. Orbiting around the nucleus are much smaller, negatively charged particles called electrons. Electrons are much smaller than protons or neutrons and have a mass of only 1/1845 of that of a proton, but each electron has an electrically negative charge the same strength, but opposite polarity to the charge on a proton. This structure means that for every proton there is an electron and as they both have equal and opposite electric charges the electrical charges balance out and the atom is electrically neutral, that is, overall neither positive nor negative.
Ions
It is possible for an atom to gain or lose electrons, and when this happens materials become electrically charged. This is what happens when a plastic pen or balloon is rubbed on certain fabrics. Electrons are ‘rubbed off’ the balloon by friction and the balloon becomes electrically charged.
When an atom has an imbalance of protons and electrons it will try to get back into balance by attracting electrons from other atoms in the vicinity, or if it has more electrons than protons it will try to share its surplus electrons with any neighbouring atoms that are short of electrons. An atom that is short of electrons is called a positive ion, and one which has too many electrons is called a negative ion. An ion is simply an atom in which the numbers of protons and electrons are unequal.
It is this action of positive and negative ions exchanging electrons to cancel out their electric charges, which makes small pieces of paper etc. stick to the electrically charged balloon. Notice that, after a while the paper falls off the balloon. This happens because eventually the atoms in the balloon and in the paper have exchanged enough electrons to lose most of their electrical attraction. In some cases this exchange of electrons is surprising, such as getting a small electric shock on touching a lift (elevator) button after walking across a carpet - an electric charge builds up on a person due to the friction between their shoes and the carpet fabric. Consequently a current flows when the metal (and electrically neutral) lift button is touched. In other examples the effect can be devastating, for example a lightning strike, where giant clouds of water droplets become charged to fantastically high voltages by friction, as they are churned up by air currents, then discharge suddenly when the insulation between the cloud and the ground (the air), breaks down.
Figure 1.0.2. Two ways of Illustrating The Silicon Atom
Silicon Atom
Figure 1.0.2 Illustrates the structure of an atom of silicon, a very common element widely used in electronics. Figure 1.0.2(a) shows how the basic (simplified) structure of the atom may be imagined, with a nucleus consisting of 14 protons (shown in red) and 14 neutrons (shown in black), surrounded by 14 (blue) electrons orbiting the nucleus in a number of ‘electron shells’. A pictorial diagram like this gives an understandable impression of the atom, a system not unlike a planetary one with orbiting satellites. However in this view it is not easy to show important features of any but the simplest atoms. Different branches of science illustrate atoms in different ways depending on what features are important to the explanation being illustrated. In electronics a simplified ‘Schematic’ diagram like that in Figure 1.0.2(b) is often used. Here the electrically important characteristics of the silicon atom are shown. The 14 electrons in their various shells with the 14 protons of the nucleus, are shown simply as one large ball having a positive charge of +14, balancing the 14 negative charges of the electrons. Because the Neutrons have no electric charge they are ignored.
Figure 1.0.2(b) shows a 2 dimensional view of the electrons orbiting the nucleus in a strict pattern of electron shells. Any particular electron can move around the nucleus in 3 dimensions and can at any time be at any point around the nucleus, rather like a satellite orbiting the earth. However it must orbit at a certain distance from the nucleus. To orbit further out from the nucleus would mean that the electron must gain energy from somewhere, and to orbit closer to the nucleus would mean that the electron would have to lose energy to somewhere.
Electron Shells
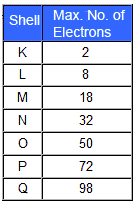
Figure 1.0.3
Electron Shells
Each electron shell is a fixed distance from the nucleus, between each shell is a ‘forbidden gap’. For an electron to move to another shell it must gain or lose sufficient energy to move to the next shell in one ‘jump’. When an electron loses energy and jumps to a lower shell (energy level) the energy may be given out as heat or light; it is this loss of energy as light that makes a light emitting diode (LED) glow.
There are up to seven electron shells around an atom and are named using the letters K (for the shell closest to the nucleus) L, M, N, O, P and Q. Some atoms (e.g. hydrogen and helium) have only one shell (the K shell) whereas very complex atoms such as uranium have electrons in all seven shells. The maximum number of electrons each shell can hold is also fixed as shown in Fig. 1.0.3.
The total number of electrons in a particular atom will equal the number of protons and is constant for each element. The distribution of the electrons between shells is also always the same for any particular element. Each shell can hold less electrons than its maximum number, but not more.
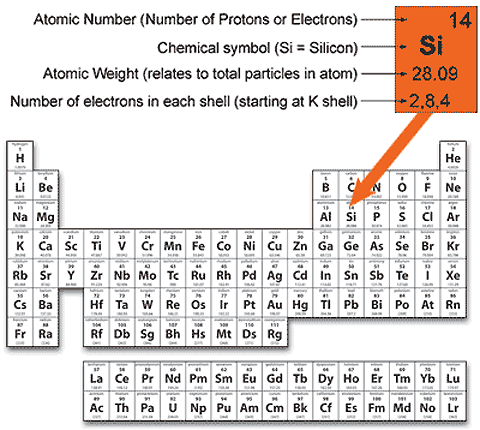
Figure 1.0.4. Silicon described in a Periodic Table
Electrons per Shell
The distribution of protons, neutrons and electrons in each of the elements found in nature is given in a Periodic Table of Elements (Fig.1.0.4). Many different versions of this table can be found in textbooks on physics and science. It is a table setting out the important properties of all known elements, mostly natural, but some man-made elements too. The elements are usually arranged in sequence beginning with hydrogen, which has the smallest atomic weight (atomic weight is a measure of the total mass of all the particles in an atom) and the simplest structure, and ending with a series of elements (some man made) which are very complex and have the largest number of particles in their structure. Each element is illustrated by the information in one box within the table. Of particular interest to electronics engineers are the numbers of electrons in the various electron shells, given at the bottom of each box. Silicon, for example has the maximum number of 2 electrons in its K shell, 8 in its L shell, but only 4 out of a possible 18 electrons in its (outer) M shell. These outer electrons are also called valence electrons and it is these electrons that can be made to migrate from atom to atom when an electric current flows through the silicon. (The inclusion of the number of electrons per shell is often omitted in periodic tables but is useful for electronics purposes).
The Electrical Properties of Materials
Any material can be considered as belonging to one of three groups, depending on its ability to pass an electric current.
1. Conductors pass current readily.
An electric potential applied across a conducting material will cause valence electrons to become detached from their atoms and move (as they are negatively charged) towards the positive potential. As an electron leaves its atom, that atom is of course deficient of negative charge and becomes a positively charged ion. This makes it attractive to another electron which takes the place of the one just attracted away, and so on. Thus a flow of electrons moves, atom by atom towards the positive terminal, being replaced by an excess of electrons at the negative terminal.
The effects of temperature on a conductor.
Heating a conductor causes a vibration within the atomic structure of the material, and this vibration ‘shakes loose’ some of the more easily detached electrons allowing them to migrate randomly from atom to atom. As temperature rises within the conductor, the vibration of the atomic structure increases, and so there are more free electrons being shaken loose from their orbits. This gives rise to more collisions between the randomly moving electrons and the electrons moving under the influence of the electric potential. This makes the flow of electrons towards the positive electrical potential more difficult. Therefore the resistance of a conductor slightly increases as its temperature rises.
In a conductor there are a large number of easily detached electrons within the structure. Some of these electrons are already moving randomly about the structure due to thermal energy (heat) at room temperature, and are called free electrons. Applying an electric potential across the conductor greatly increases the number of free electrons as they move from the negative to positive terminal as previously described. As these free electrons travel through the structure however, they are bound to collide with some of the electrons moving under the influence of heat, as well as being attracted to individual atoms as they jump from one atom´s valence shell to the next. It is these collisions and the pull of individual atomic nuclei, which give the material the electrical property of resistance. The resistance of a material indicates how well or how badly the material conducts (i.e. allows the movement of electrons under the influence of an electric potential or voltage). The higher the value of resistance, the more difficult it is to move the electrons. Materials with higher resistance will therefore need a higher potential (higher voltage) applied to cause a given amount of current, (movement of electrons).
2. Insulators
An insulator has all its electrons tightly bonded to the nucleus and so it takes very large forces of either heat or potential to dislodge them. Insulators do not normally pass current. In some cases, for example at high temperatures or with very high voltages applied, some insulating materials will conduct. In these circumstances the insulating material is said to have ‘Broken down’ and usually the structure of the material is permanently damaged. In some insulators (glass for example) heating the material to a high temperature will vibrate the atoms so violently that enough electrons will be shaken free for conduction to occur. Allowing the glass to cool again stops conduction. In most insulators however, conduction, whether caused by excessive heat or by excessive voltage, will permanently destroy the material. For this reason any material used for electrical insulators, will have a safe working limit quoted by the manufacturer of the insulator,for both voltage and temperature.
3. Semiconductors
Semiconductors fall between groups 1 and 2. They do not normally pass current easily at room temperature, having a resistivity higher than conductors, but lower than insulators. They have properties however which make them very useful for the manufacture of electronic devices such as transistors and diodes.
The difference between the three groups of materials lies in the number of easily detached electrons within the atomic structure. The electrons concerned are more or less loosely held in the outermost electron shells called valence shells and so these electrons are called valence electrons.
In a semiconductor the bond between the valence electrons and their nucleus is much stronger than in a conductor, so far fewer electrons are free to move when a potential is applied. When current does flow, the chances of a collision between the electrons moving due to the electric potential, and randomly moving electrons due to heat, is much less. Additionally the heating of the material frees electrons previously held by their atoms and so these electrons are also free to add to the current flow. Although of course the total current flow is much less than in a conductor, the amount by which current flow increases due to heat is proportionally greater. In use therefore, it is important to keep components made from semiconductor materials, such as transistors, cool. Otherwise an effect called thermal runaway can occur. This is when an increase in temperature causes an increase in current, which in turn causes a further increase in temperature, leading to further increasing current. A process that can escalate until the material passes so much current it is destroyed.

